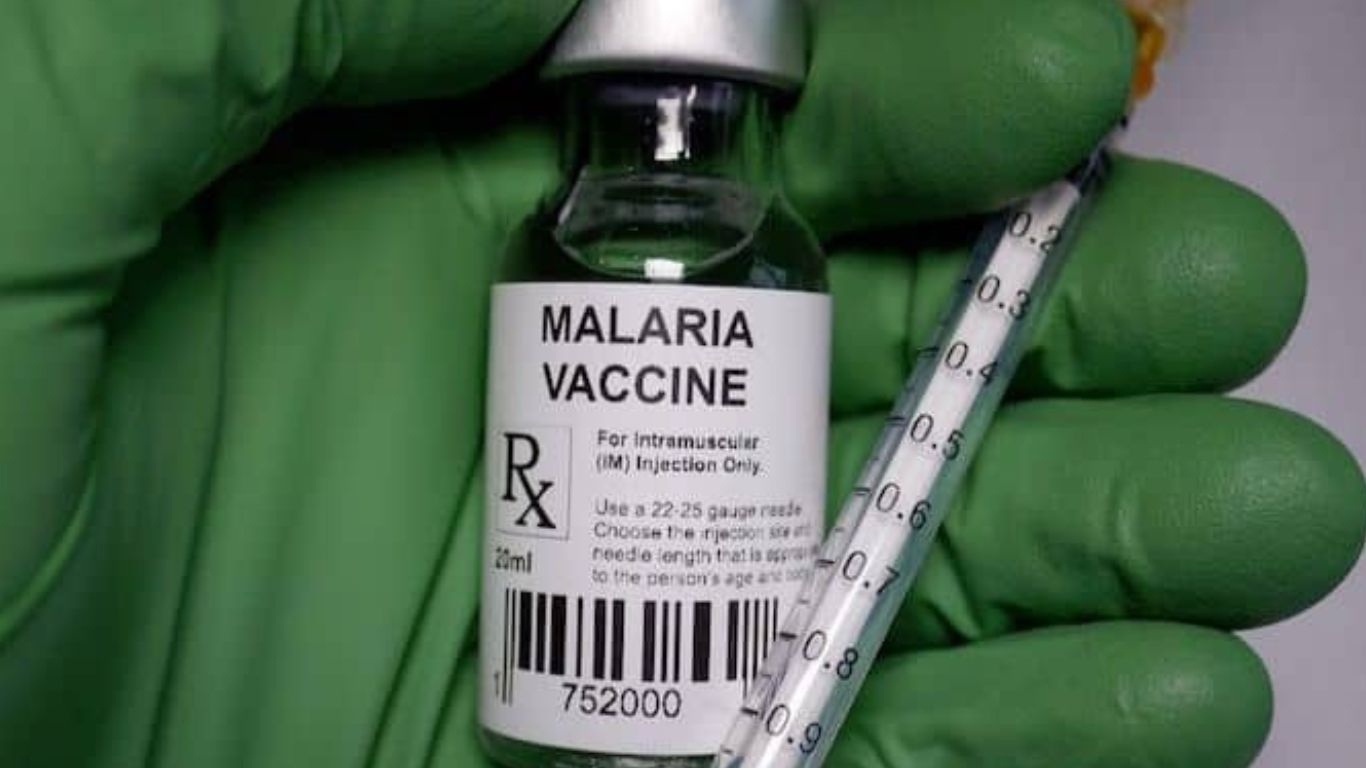
The National Agency for Food and Drug Administration and Control, NAFDAC announced conditional approval of the vaccine in Abuja on Monday, 17th April, 2023.
The approval of the new R21 malaria vaccine by NAFDAC has made Nigeria to become the second country in Africa after Ghana to approve the R21/Matrix-MTM malaria vaccine, which is hoped to save millions of lives in the country.
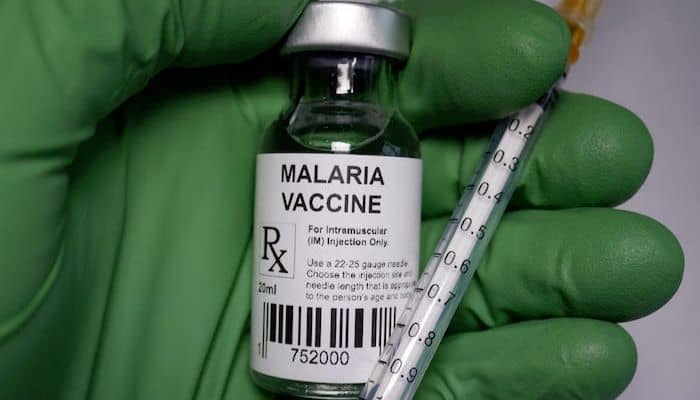
The DG of NAFDAC, Mojisola Adeyeye, disclosed this at a press briefing in Abuja on Monday, 17th April, 2023, that the vaccine prevents clinical malaria in children between 5 to 36 months and provides 71 percent protection.
The DG named Fidson healthcare limited as the market authorization holder. She further explained that the approval of the vaccine follows a stringent review of the dossier of the vaccine by the agency and critical stakeholders in line with the World Health Organisation standards.
Mrs. Mojisola Adeyeye, added that the conditional approval is as a result of the huge burden of malaria in Nigeria with children being the most vulnerable.
Story: Ezekiel Kefas